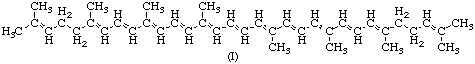
Carotenoids are a class of hydrocarbons (carotenes) and their oxygenated derivatives (xanthophylls) consisting of eight isoprenoid units joined in such a manner that the arrangement of isoprenoid units is reversed at the centre of the molecule so that the two central methyl groups are in a 1,6-positional relationship and the remaining nonterminal methyl groups are in a 1,5-positional relationship. All carotenoids may be formally derived from the acyclic C40H56 structure (I below), having a long central chain of conjugated double bonds, by (i) hydrogenation. (ii) dehydrogenation, (iii) cyclization, or (iv) oxidation. or any combination of these processes.
This class also includes certain compounds that arise from certain rearrangements of the carbon skeleton (I), or by the (formal) removal of part of this structure.
For convenience carotenoid formulae are often written in a shorthand form as
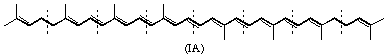
(General formula; broken lines indicate formal division into isoprenoid units).
Rule Carotenoid 2. The stem name
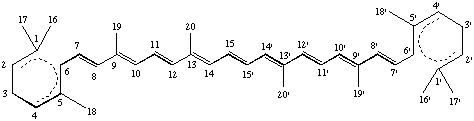
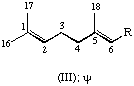
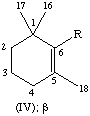
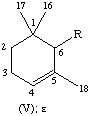
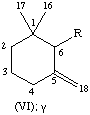
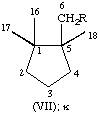
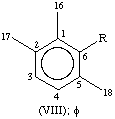
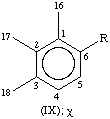
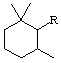
All specific names are based on the stem name 'carotene', which corresponds to the structure and numbering shown in II, where the broken lines at the two terminations are intended only to represent two 'double-bond equivalents'. Individual compounds may have C9 acyclic end groups with two double bonds at positions 1,2 and 5,6 (e.g., III) or cyclic end groups (such as IV, V, VI, VII, VIII and IX).
Rule Carotenoid 3. Specific names; End-group designations
3.1. The name of a specific carotenoid hydrocarbon is constructed by adding two Greek letters as prefixes to the stem name 'carotene' (defined in Rule Carotenoid 2), these prefixes being characteristic of the two C9 end groups.
3.2. The prefixes are
| Type | Prefix | Formula | Structure |
| Acyclic | ψ | C9H15 | III |
| Cyclohexene | β, ε | C9H15 | IV, V |
| Methylenecyclohexane | γ | C9H15 | VI |
| Cyclopentane | κ | C9H17 | VII |
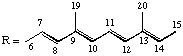
| Aryl | φ, χ | C9H11 | VIII, I |
 |  |  |
 |  |  |
 |  | |
3.3. The Greek-letter prefixes are cited in alphabetical order; the first is separated from the second by a comma, and the second is connected to the stem name by a hyphen.
Note: The Greek-letter alphabetical order is β (beta), γ (gamma), ε (epsilon), κ (kappa), φ (phi), χ (chi), ψ (psi).
Examples:

ε,ε-Carotene
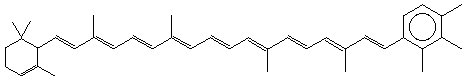
ε,χ-Carotene
Notes:
(i) The Greek-letter prefixes are derived: β and ε from the symmetrical carotenoids with the trivial names 'β-carotene' and 'ε-carotene', κ from the symmetrical capsorubin, γ and φ from γ- and φ-ionone, φ for phenyl, and χ, the next Greek letter after φ.
(ii) α and δ, from the trivial names α- and δ-carotene, are not used.
3.4. When, in a modified or degraded carotenoid, an end group can be derived from more than one specific end group, the end group chosen as the basis of the name is that occurring earliest in the alphabetical order (see Rule Carotenoid 3.3).
Example:
is a derivative of the β end group, not the ε end group.
Rule Carotenoid 4. Numbering of carotenoid hydrocarbons
The basic system of numbering is that shown in structure II of Rule Carotenoid 2; the end groups are numbered as indicated in Rule Carotenoid 3. If the two end groups are dissimilar, lower (unprimed) numbers are given to that end of the molecule which is associated with the Greek-letter prefix cited first in the name. [All unprimed locants are cited before primed locants (ref 1)]
Examples:
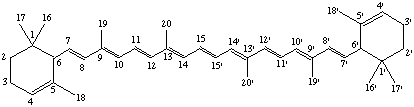
ε,ε-Carotene

ε,χ-Carotene
Note: It is recommended that formulae be drawn so that unprimed numbers are on the left-hand side.
Rule Carotenoid 5. Nor carotenoids and seco carotenoids
5.1. Nor Carotenoids
Elimination of a CH3, CH2, or CH group from a carotenoid is indicated by the prefix "nor", which in all cases is preceded by the locant of the carbon atom that has been eliminated. The prefix is nondetachable. When alternatives are possible, the locant attached to nor is the lowest possible (ref 2). The basic numbering of the carotenoid is retained in the nor carotenoid.
Examples:
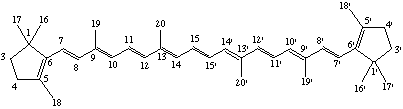
2,2'-Dinor-β,β-carotene
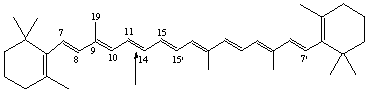
(12,13,20 formally 'removed' at the marked point)
12,13,20-Trinor-β,β-carotene
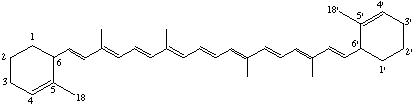
16.17,16',17'-Tetranor-ε,ε-carotene
5.2. Seco Carotenoids
Fission of the bond between two adjacent carbon atoms (other than carbon atoms 1 and 6 of a cyclic end group) with addition of one or more hydrogen atoms at each terminal group thus created, is indicated by the prefix "seco", the original carotenoid numbering being retained.
Example:
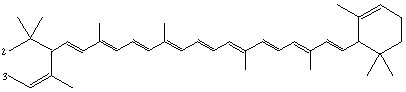
2,3-Seco-ε,ε-carotene
Rule Carotenoid 6. Changes in hydrogenation level
Carotenoid hydrocarbons, including carotenoid acetylenes and allenes, that differ in hydrogenation level from the corresponding carotenoid hydrocarbon defined by Rule Carotenoid 3, are named from the latter by use of the prefixes "hydro" and "dehydro" together with the locants specifying the carbon atoms at which hydrogen atoms have been added or removed.
These prefixes are nondetachable (ref 3) and immediately precede the Greek-letter prefixes denoting the end groups, and, if both occur in one name, are cited in the order: dehydro before hydro (multipliers do not affect the order).
Example: tetradehydrodihydro
Note: Since, to maintain valency requirements, hydrogen atoms are, formally, always added or removed in pairs these prefixes will always be used with an even-number multiplier, e.g. tetrahydro, didehydro.
Examples:
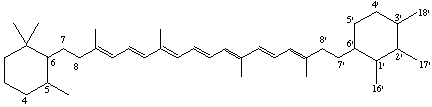
5,6,7,8,1',2',3',4',5',6',7',8'-Dodecahydro-β,χ-carotene
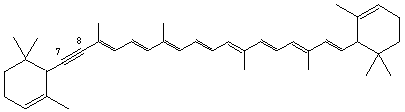
7,8-Didehydro-ε,ε-carotene
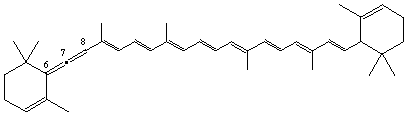
6,7-Didehydro-ε,ε-carotene
Rule Carotenoid 7. Oxygenated derivatives
7.1. Oxygenated (and other) derivatives of carotenoid hydrocarbons are named by use of suffixes and prefixes according to the rules of general organic chemical nomenclature (ref 4).
Of the oxygen-containing characteristic groups present, that occurring earliest in the sequence carboxylic acid, ester of carotenoid acid, aldehyde, ketone, alcohol, ester of carotenoid alcohol, is chosen as principal group [following IUPAC Rule C-10.3 [see ref 1)] and is cited by use of a suffix; all other groups are cited as prefixes.
Examples:
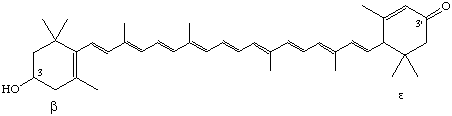
3-Hydroxv-β,ε-caroten-3'-one
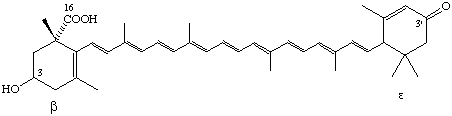
3-Hydroxy-3'-oxo-β,ε-caroten-16-oic acid
7.2. A nonbridging ether group is named by use of the appropriate alkoxy or aryloxy prefix [Rule C-211.2 (see ref 1)].
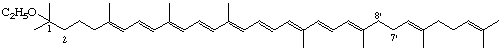
1-Ethoxy-1,2,7',8'-tetrahydro-ψ,ψ,-carotene
7.3. Oxygen bridges are indicated by use of the prefix 'epoxy'; this prefix is preceded by the locants of the two carbon atoms that form the bridgeheads of the oxygen bridge.
Note: The prefix 'epoxy' denotes replacement, by an oxygen bridge, of a hydrogen atom at each of two carbon atoms already otherwise connected to one another. An epoxide, notionally formed by adding an oxygen atom to a double bond, is therefore an epoxydihydro derivative of the original compound.
Examples:
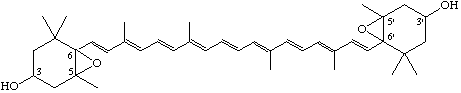
5,6:5',6'-Diepoxy-5,6,5',6'-tetrahydro-β,β-carotene-3,3'-diol

5,8:5',8'-Diepoxy-5,8,5',8'-tetrahydro-β,β-carotene
7.4. Compounds that may be formally derived from a carotenoid hydrocarbon by the addition of the elements of water (H, OH) or of methanol (H, OCH3) to a double bond are named as 'hydroxydihydro' or 'methoxydihydro' derivatives.
Examples:
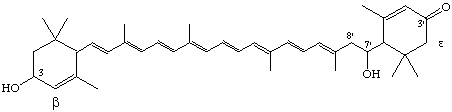
3,7'-Dihydroxy-7',8'-dihydro-β,ε-caroten-3'-one
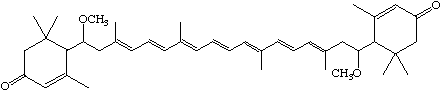
7,7'-Dimethoxy -7,8,7',8'-tetrahydro-ε,ε-carotene-3,3'-dione
1. IUPAC Nomenclature of Organic Chemistry (Pure Appl. Chem. 11, Nos. 1 and 2 (1965); Sections A, B and C published by Butterworths, London, 1971) indicates that, in ranking locants for priority, a primed numeral ranks immediately after the same numeral unprimed. In general organic nomenclature the common practice is therefore to cite locants in the order x, x', (x+n), (x+n)'. The established sequence in the carotenoid field is, however, to cite an unprimed numerals before any primed numerals, and this practice is followed in these rules. [Now included in the 1979 edition with sections D, E, F and H]
2. The contrast with steroid usage [IUPAC-IUB 1971 Definitive Rules for Steroid Nomenclature, Pure Appl. Chem. 31, Nos. 1-2 (1972)] Rule 2S-6, where the prefix 'nor' is associated with the highest permissible number, is to be noted. [Now as the 3rd edition, 1989.] The 1981 IUPAC-IUB Nomenclature of Retinoids recommended the use of the higher number to follow IUPAC Section F; Natural Product Nomenclature, and proposed that when the carotenoid rules are next revised this should be used there too.
3. IUPAC Nomenclature of Organic Chemistry (see ref 1), Rule C-16.1. allows the prefix 'hydro' to be detachable or nondetachable and the former has become the established usage in general organic chemistry. However, the common practice in carotenoid names is now to use this prefix as nondetachable, a practice that is followed in this set of rules.
4. IUPAC Rules, Nomenclature of Organic Chemistry (see ref 1), Subsections C-2 to C-4.